Research Interests
Wide-bandgap Oxides
This class of materials is often also referred to as transparent conducting oxides (TCOs), as they combine a large bandgap (and thus optical transparency) with dopability (and thus conductivity). Their large bandgap also makes them of interest for high-power devices.
Our group is currently working on 3 research projects within this category:
1) Optimizing oxygen vacancy transport in memristors
This project is funded by a NSF Future of Semiconductors (FuSe2) grant (DMR-2425549) and tries to optimize Al2O3/Ga2O3/MgO-based memristors grown using Atomic Layer Deposition (ALD). Oxygen vacancies form conducting filaments in these memristors, and the creation or destruction of the filaments by applying positive or negative bias voltages leads to a switch between high-resistive (off) and low-resistive (on) states. This switching occurs with significant hysteresis, which mimics the activation of biological neurons, and which leads to memristor behavior. Our calculations focus on elucidating the atomic scale processes. For example, we showed that doping with Mg leads to higher resistivity in the off state, while at the same time increasing the number of oxygen vacancies.
Representative recent publications:
- A. Marshall, R. Goul, B. Dodson, R. Sakidja, H. Peelaers, S. Seacat, K. Bray, D. Ewing, M. Walsh, and J. Z. Wu "Probing the Effects of the First Atomic Layer on the Dynamic Behavior of Sub-2 Nm MgO/Al2O3 Memristors", ACS Applied Materials & Interfaces 17, 49930 (2025) [doi:10.1021/acsami.5c04654].
- Aafiya, A. Marshall, B. Dodson, R. Goul, S. Seacat, H. Peelaers, K. Bray, D. Ewing, M. Walsh, and J. Z. Wu "Probing Electronic and Dielectric Properties of Ultrathin Ga2O3/Al2O3 Atomic Layer Stacks Made with in Vacuo Atomic Layer Deposition", Journal of Applied Physics 136, 025104 (2024) [doi:10.1063/5.0208590].
- R. Goul, A. Marshall, S. Seacat, H. Peelaers, F. C. Robles Hernandez, and J. Z. Wu "Atomic-Scale Tuning of Ultrathin Memristors", Communications Physics 5, 1 (2022) [doi:10.1038/s42005-022-01037-4].
2) Optimizing Ga2O3-based high-power devices
Ga2O3 is of interest for high-power devices because of its ultra-wide bandgap (~4.8 eV), which is even larger than that of SiC and GaN, the current leading materials for power electronics. This large bandgap translates to a very high breakdown electric field, estimated to be even larger than that of GaN and SiC. This fundamental property means that Ga2O3-based power devices can, in theory, block higher voltages with lower resistance than existing technologies, leading to more efficient power conversion and management in applications like electric vehicles, fast chargers, and grid-scale power systems. An additional and significant practical advantage is that high-quality, large-diameter single-crystal wafers can be grown from the melt using methods similar to those for sapphire or silicon, which promises a much lower cost compared to the more complex manufacturing processes for SiC and GaN wafers.
Our group has been very active in providing an in-depth understanding of the material properties of Ga2O3, and on ways to modify and optimize these properties. For example, we have extensively studied alloys based on Ga2O3, demonstrating how alloying with Al2O3 or In2O3 can be used to tune the band gap and engineer heterostructures. Recently, we predicted that monoclinic (In,Al)2O3 alloys are ideal to form heterostructures with Ga2O3, as these can be lattice-matched while providing a large (1 eV) conduction-band offset for carrier confinement. Most recently, in a new arXiv preprint, we uncovered the origin of the long-standing mystery of poor n-type doping in high-Al content (Al,Ga)2O3 alloys, showing that cation vacancies form and compensate the intentional donors.
Representative recent publications
- S. Seacat, J. L. Lyons, and H. Peelaers "Properties of Orthorhombic Ga2O3 Alloyed with In2O3 and Al2O3", Applied Physics Letters 119, 042104 (2021) [doi:10.1063/5.0060801].
- S. Seacat, J. L. Lyons, and H. Peelaers, "Computational design of optimal heterostructures for beta-Ga2O3", Physical Review Materials 8, 014601 (2024) [doi:10.1103/PhysRevMaterials.8.014601].
- S. Seacat and H. Peelaers, "Achieving n-type doped monoclinic (InxAl1-x)2O3 alloys", Journal of Applied Physics 135, 235705 (2024) [doi:10.1063/5.0211069].
- S. Seacat and H. Peelaers, "Origin of donor compensation in monoclinic (AlxGa1-x)2O3 alloys", arXiv:2602.02879 (2026) [https://arxiv.org/abs/2602.02879].
3) Exploring V2O5 as cathode material for non-Li-ion batteries
This research is supported by an NSF CAREER award (DMR-2339751) and has as goal to use first-principles calculations to understand the basic material properties of V2O5 and its many polymorphs. So far, we focused on the electronic and structural properties of the single- and double-layered polymorphs. We found that the electronic properties are remarkably similar, despite the large structural differences between the polymorphs. We also elucidated that the electronic transport takes place through polaron hopping, and figured out the minimum-energy pathways for this hopping in the various polymorphs.
Representative recent publications
- "Electronic and structural properties of V2O5 layered polymorphs", arXiv:2603.04193 (2026) [https://arxiv.org/abs/2603.04193]
Organic/organic and organic/inorganic interfaces
Our group explores fundamental physics at organic and hybrid interfaces, supported by the Department of Energy (DOE DE-SC0024525). Using first-principles calculations, we study how the structure and composition of these interfaces govern critical processes such as charge separation, energy level alignment, and carrier dynamics. For example, our work on organic/organic interfaces has revealed that near-zero energy offsets can still enable efficient charge separation, challenging conventional design rules for organic semiconductors. In organic/inorganic systems, we have shown that molecular layers on two-dimensional (2D) materials can create nanoscale periodic trapping sites for interlayer excitons, and that the electrostatic potential in 2D crystals can be modulated by arrays of polar molecules. We have also elucidated the mechanisms of ultrafast charge transfer in organic-TMD heterostructures, demonstrating how energy valley-dependent processes govern carrier dynamics. This research provides fundamental insights for designing next-generation optoelectronic, photovoltaic, and quantum devices that leverage the tunability of both organic and inorganic components.
Representative recent publications:
- N. Fuller, K. Rijal, E. Udeh, S. Amos, H. Peelaers, and W. Chan "Charge Separation at Organic Interfaces with Near-Zero Energy Offset: A Step toward Designing Inorganic-like Organic Semiconductors", Journal of Physical Chemistry Letters 16, 12545 (2025) [doi:10.1021/acs.jpclett.5c02996].
- N. Fuller, F. Rudayni, S. Amos, K. Rijal, S. A. Maroufian, P. Valencia-Acuna, T. Karl, H. Zhao, H. Peelaers, Q. Zhou, and W. Chan "Modulation of Electrostatic Potential in 2D Crystal Engineered by an Array of Alternating Polar Molecules", Nano Letters 24, 10258 (2024) [doi:10.1021/acs.nanolett.4c02555].
- K. Rijal, S. Amos, P. Valencia-Acuna, F. Rudayni, N. Fuller, H. Zhao, H. Peelaers, and W. Chan "Nanoscale Periodic Trapping Sites for Interlayer Excitons Built by Deformable Molecular Crystal on 2D Crystal", ACS Nano 17, 7775 (2023) [doi:10.1021/acsnano.3c00541].
- P. Valencia-Acuna, S. Amos, H. Peelaers, and H. Zhao "Energy-Valley-Dependent Charge Transfer in Few-Layer Transition Metal Dichalcogenide Heterostructures", Physical Review B 108, 085302 (2023) [doi:10.1103/PhysRevB.108.085302].
- P. Valencia-Acuna, T. R. Kafle, P. Zereshki, H. Peelaers, W. Chan, and H. Zhao "Ultrafast Hole Transfer from Monolayer ReS2 to Thin-Film F8ZnPc", Applied Physics Letters 118, 153104 (2021) [doi:10.1063/5.0045710].
Research Highlights
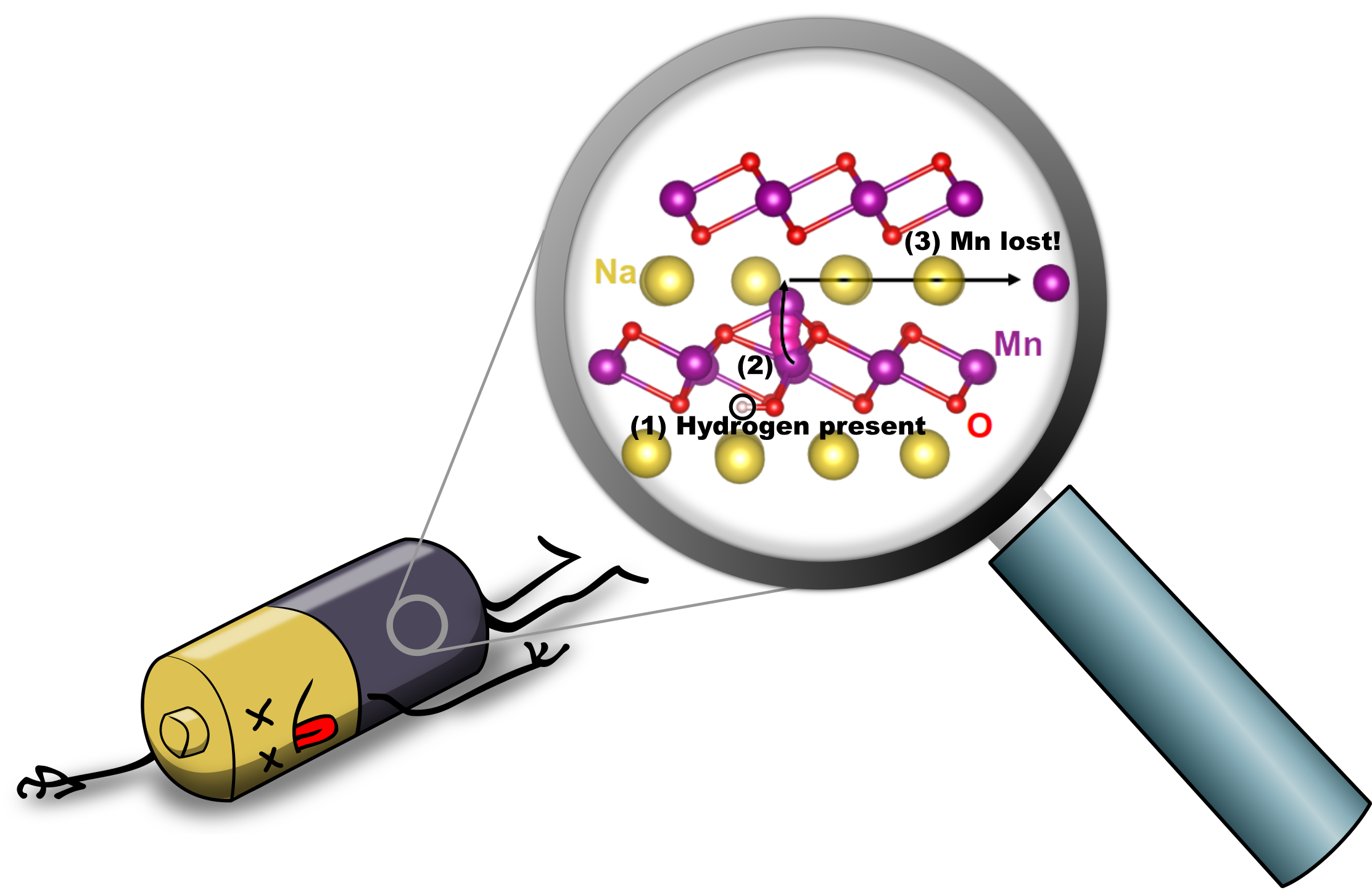
Hydrogen-Induced Degradation of Battery Cathodes
Batteries power our lives: we rely on them to keep our cell phones and laptops buzzing and our hybrid and electric cars on the road. Ever more widespread adoption of the most commonly used lithium-ion batteries may actually lead to increased cost and potential shortages of lithium—which is why sodium-ion batteries are being intensely researched. Their performance is good, and sodium is cheap and abundant; however, the sodium-ion batteries have shorter lifetimes than their lithium-based siblings. Our research revealed the presence of hydrogen as the culprit. Read more details at phys.org, sciencedaily, The UCSB Current, and several other locations.
Transparent Conducting Oxides
Transparent conducting oxides (TCOs) are a technologically important class of materials with applications ranging from solar cells, displays, smart windows, and touch screens to light-emitting diodes. TCOs combine high conductivity with transparency in the visible region of the spectrum. A fundamental understanding of the factors that limit transparency in TCOs is essential for further progress in materials and applications. Direct transitions do not lead to absorption of visible light. However, indirect processes, such as those mediated by phonons, can lead to absorption. Using calculations I showed the size and nature of the fundamental limit of the transparency of SnO2. This important result received wide spread attention: by the editors of Applied Physics Letters (selected as a research highlight and as Editor's Pick for the journal's 50th Anniversary), by the scientific community (7th most read article in January 2013), by the scientific blogosphere (e.g., physorg.com, photonics.com, sciencedaily.com,...), by the trade press (Photonics Spectra), and by the regular press (Santa Barbara News-Press).
Artist’s impression of key results. Three beams of light (red for infrared, yellow for visible light, and violet for ultraviolet) travel through a layer of SnO2. Absorption by the conduction electrons in the oxide reduces the intensity of the beams. The calculations showed that absorption is 5 times stronger for ultraviolet and 20 times stronger for infrared, compared to absorption of visible light. There is no sound in this video. |
Layered Materials
Layered materials show a lot of promise as materials for a wide range of devices, as evidenced by the 2010 Nobel Prize in Physics awarded for graphene, a single layer of carbon atoms. However, graphene lacks a band gap, hindering effective device applications. One possible approach I studied to open a gap is by bonding fluorine atoms to a graphene layer, called fluorographene. Other two-dimensional materials are also highly promising. Recent experiments on molybdenum disulfide (MoS2) showed that it could be used in FETs. However, a lot of crucial information about the band structure and electronic properties of this material is still lacking, hampering interpretation of experiments and preventing accurate device modeling. I have therefore calculated key materials parameters such as band gaps and effective masses, as well as performed an investigation of the effects of strain. I showed how strain allows engineering the nature (direct vs indirect) and size of the band gap and the magnitude of effective masses. In addition, insight into the fundamental physics was obtained by considering the transition between the bulk and the monolayer. This work was featured on the National Institute of Computational Sciences (NICS) website.
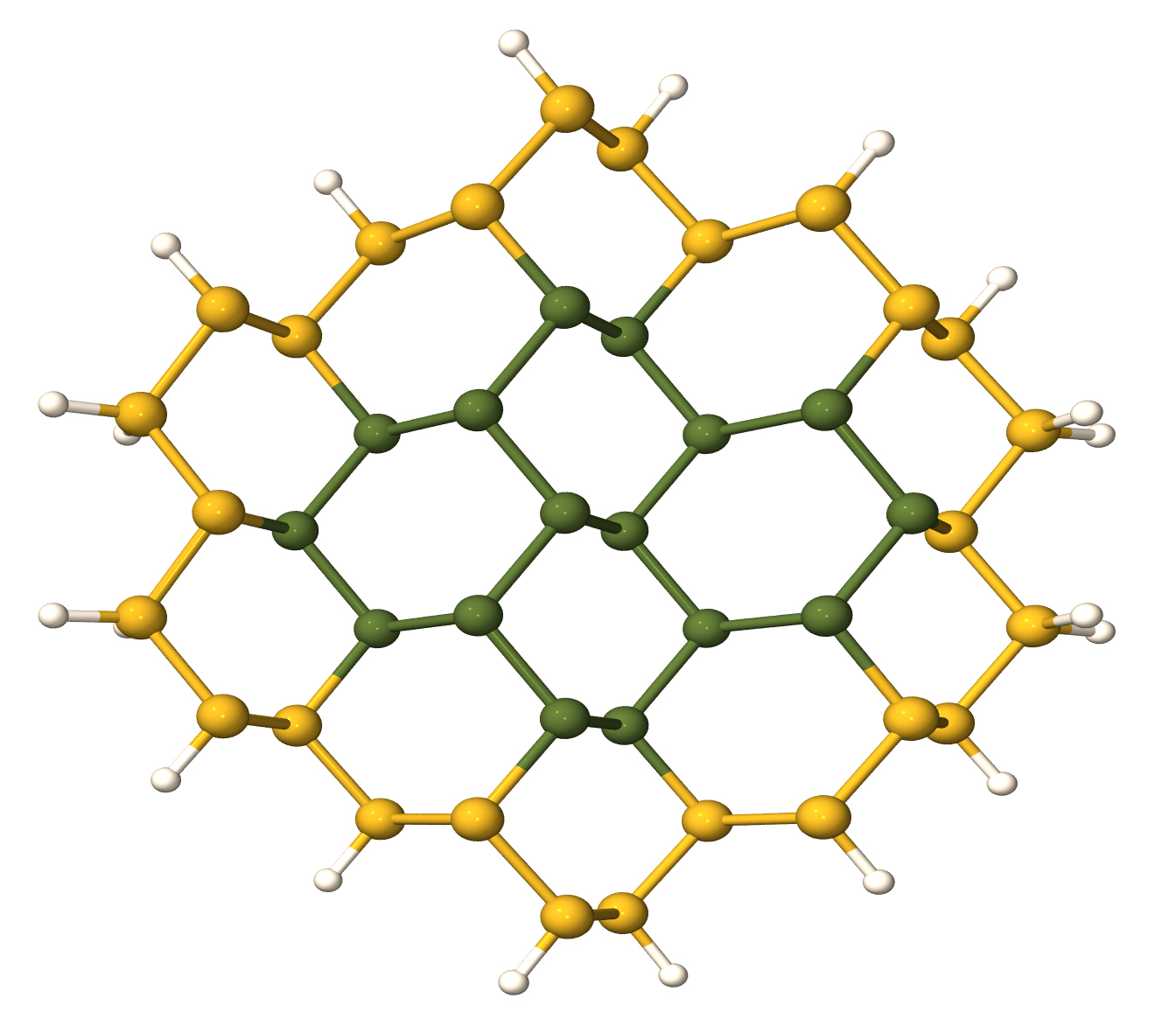
Semiconducting Nanowires
Nanotechnology is becoming increasingly important for future applications. Nanowires form a possible nanoscale building block and applications in electronics have already been demonstrated, e.g. for field-effect transistors (FET). FETs are building blocks of the semiconductor industry, and it was shown that nanowires have device properties that can exceed the performance of the current technology. Another promising field is the use as sensors for biological and chemical substances; applications as highly sensitive sensors for virus particles and cancer markers have already been shown. Control of dopant incorporation is essential for these applications. I have performed detailed studies, using a variety of computational techniques, of boron and phosphorus dopants in silicon and germanium, and silicon/germanium core-shell nanowires. The results have allowed me to explain the experimentally observed conductance difference between B-doped and P-doped nanowires, and I have predicted that P dopants tend to segregate to the surface of the nanowires, a finding that has been confirmed experimentally.